The Ongoing Battle: My Personal Fight Against the PBM Machine
- Mar 16
- 8 min read
First, Let Me Tell You What I'm Fighting For
Before I get into the timeline of what happened, I want to explain why this particular fight was not just frustrating, it was existential.
I have a form of Primary Immunodeficiency called Common Variable Immune Deficiency, or CVID. In plain terms, my immune system does not produce enough Immunoglobulin G (IgG), the antibodies that your body uses to identify and fight off infections. Most people manufacture IgG naturally. My body cannot do this adequately. Without it, even routine infections can become life-threatening.
The only treatment for CVID is lifelong Immunoglobulin Replacement Therapy (IRT), infusions of IgG antibodies pooled from the donated plasma of 1,000-15,000 individual donors per batch. This is not a medication with a generic version or a biosimilar alternative. It is not a drug your doctor can swap for something cheaper from a preferred tier. There is no therapeutic substitute. The medication I was fighting for was the only type of treatment for my condition that exists.
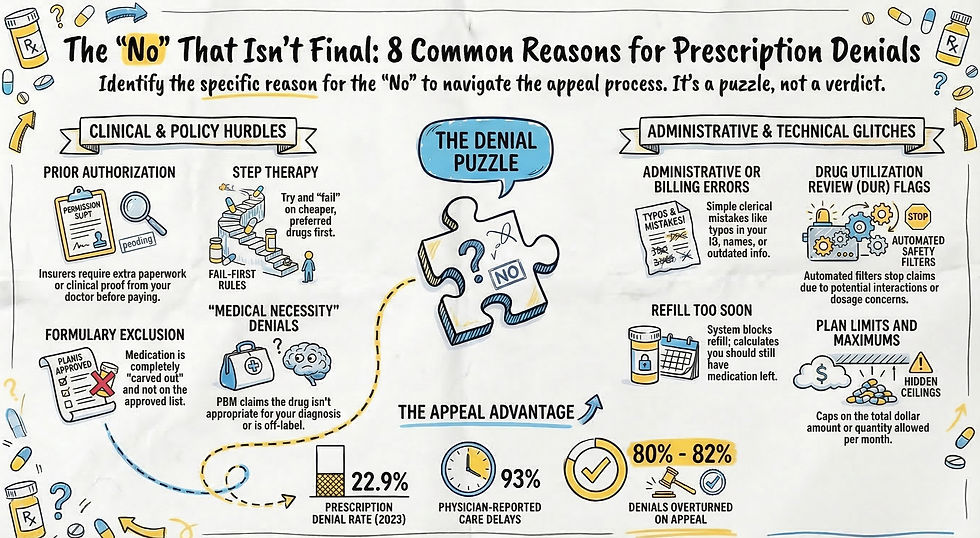
This matters because the PBM's standard playbook — step therapy, preferred alternatives, fail-first requirements — had no clinical basis when applied to my situation. You cannot "try a cheaper drug first" when no biosimilar alternative drug exists.

Week One: The First "No"
The denial arrived the way they always do, first a text, then a portal message, and finally that dreaded plain white envelope, with dense language designed to sound final. After more than two years of stability on this medication, my reauthorization had been denied. The stated reason was all too familiar. The PBM stated that my plan only covers my prescribed medication when I meet ONE of these options A) try one of their preferred drugs and show that it does not work for me, OR B) My doctor gives a medical reason as to why I cannot take their preferred drug. They denied my request because the insurance company claims they have no evidence of me meeting either criteria.
I had been through enough of these battles to know that the first no is rarely the last word. I opened my digital communication log, a habit I have built over ten years of advocacy, and started making calls. What I found immediately was that the Notice of Adverse Determination said I could ask for a free copy of the benefit provision, guideline, protocol or OTHER similar criterion used to make the decision AND any OTHER information related to their decision by calling Customer Care. That seemed simple enough, but that simple request started a battle that hasn't been resolved yet!
The back of the letter stated the Plan Approved Criteria used was “Subcutaneous Immune Globulin (SCIG) ACSF SGM.” I understood the first part. SCIG is the abbreviation for my weekly infusions. A quick online search revealed that ACSF meant Advanced Control Specialty Formulary. This is the policy which focuses on managing specialty drugs by preferring lower-cost alternatives. So basically, the PBM was claiming my prescribed medication was more expensive than their preferred drug. This was easy to disprove. I looked at my previous claims for the past year for my prescribed medication to discover the breakdown of costs (My cost, the discount applied, and amount paid by insurance). I then did a price check through my insurance for their preferred drug. Turns out their drug was going to not only cost me more out of pocket, but also cost the insurance more. Why would they “prefer” that drug? One word came to mind—Rebates. This is not the type of rebate that is appreciated by the budget-conscious shopper. This rebate is when a PBM receives money back from a drug manufacturer for prioritizing a specific medication. These rebates are negotiated, often confidential, retroactive discounts based on volume, intended to increase a drug's market share in exchange for preferred status. Seems a bit shady, right?
What did that last abbreviation stand for? SGM means Specialty Guideline Management which ensures medications are used for FDA-approved indications and that patients have tried appropriate, lower-cost therapies first (step therapy). Was the insurance claiming my current prescribed medication was not FDA-approved for CVID? If that’s the case, this would be an easy thing to refute. SCIG therapy was approved by the FDA in 2006.There was no mention of the actual term “step therapy” in the denial, but the wording of “must try one preferred alternative” is the very definition of step therapy.
My Person
My doctor has a designated staff that deals with prior authorizations full-time. Unfortunately, Prior Authorizations are an extra time and financial burden to medical practices. Find out which person is going to battle for you at your doctor’s practice and get to know them. They have a stressful job dealing with insurance battles daily. My “person” is Misti. She immediately reached out to me when the denial came in. She gave me access to everything she had submitted for the prior authorization (clinical notes, labs, medical history, evidence of trying the preferred drug and having adverse events, evidence of successful on-going treatment with current prescription). Yes, you read that correctly. The doctor’s office had submitted documentation that I met not ONE but TWO of the criteria A & B that the PBM claimed they had no evidence of.
So they want me to try a preferred drug that is not a lower cost alternative and that is not biosimilar to my prescribed drug. They have evidence showing I have tried their preferred drug and had an adverse reaction. This should be easy to disprove! (I completely underestimated the willful clinical ignorance and bureaucratic gaslighting of my PBM.)
Step Therapy Exception
Misti, my doctor, and I began building the request for a step therapy exception and ultimate appeal. My doctor wrote a Letter of Medical Necessity that checked off all the boxes. It directly addressed the denial reason, cited the FDA prescribing information for my specific medication, referenced peer-reviewed research, provided my diagnosis and medical history, explained in detail that I had tried one preferred drug and how it did not work for me. He also noted the adverse events that I had experienced on their preferred drug as well as explaining my successful treatment with “my preferred drug” that my doctor had been prescribing. Side Note: It was odd to me that the PBM claimed I had not tried their drug when they had access to my prescription claims showing where I tried it (due to a forced non-medical switch by them). Also, how could they ignore the fact that they had approved my prescribed current medication for the past 2-3 years? The request was marked URGENT-Expedited Review Requested.
In addition to the LOMN and files submitted by my doctor, I submitted information to the PBM to be considered in the appeal. I invoked TN Code § 56-7-3502 — Tennessee's step therapy override provision. The PBM could not ignore that I was “..receiving a positive therapeutic outcome on a prescription drug selected by [my] healthcare provider.” I also noted in my documentation that The FDA Purple Book (an online database listing all FDA-licensed biologics) shows there are no biosimilar alternatives for my medication since subcutaneous immunoglobulin (SCIG) therapies are unique and not interchangeable. This was evidence that I should have never been forced to try the PBM’s preferred drug years ago and certainly should not have to be their experimental lab rat now!
Week Two: Bureaucratic Amnesia in Action
The 72-hour expedited window came and went without a decision. I documented every hour of the delay. When I finally reached a representative who could speak to the status of the file, I was told the appeal was "under clinical review." The same language. The same limbo.
This is where the paper trail proved its worth. I had the dates, the names, the ticket numbers. I could demonstrate that we had submitted complete documentation, that the expedited deadline had been missed, and that information was clearly being ignored. I put all of this in writing and requested a peer-to-peer review between my doctor and a board-certified immunologist — not a generalist.
The PBM scheduled the peer-to-peer call. Then "missed" it. I documented that too.
Week Three: The Circular Denial
By week three, another formal written denial had arrived. It referenced again the clinical criteria and included the standard language stating I could request the documentation used in the determination. I did exactly that. I called the PBM Customer Care line to request copies of all the documents their own denial letter stated I was entitled to. It seemed like that should be an easy enough process, but that’s when things went sideways quickly. After repeating my request like a broken record, representatives shuffled me up the line to other reps and eventually I was connected to a specialty appeals reviewer. I asked where I could access the Subcutaneous Immune Globulin (SCIG) ACSF SGM document that was referenced in the denial. The response was that the criteria were "proprietary." What? Doctors must provide time-consuming documentation, but the PBM thinks it doesn’t have to be transparent about the criteria used for making decisions.
I had seen this before. The circular denial: denied based on criteria you're not allowed to see. I called back again and put my request for information in writing, this time quoting the specific federal and state laws that require disclosure. I also drafted a formal complaint to the Tennessee Department of Commerce and Insurance (TDCI), detailing the missed expedited deadline, the refusal to disclose the denial criteria, and documentation of multiple statutes that the PBM had broken.
Filing a regulatory complaint is not a last resort. It is a power move. PBMs answer to state regulators in ways they do not answer to individual patients. The moment a complaint number exists, the dynamic changes.
February 13, 2026: The Day the System Broke Down
Four weeks in, something happened that I still find difficult to describe without shaking my head. On a single Friday afternoon, Caremark's internal systems contradicted themselves in real time — four times in six hours.
11:55 AM — Automated call: DENIED. I started planning the next appeal level.
1:09 PM — Text message: APPROVED. Relief, but also confusion.
3:21 PM — Automated call: DENIED. Anger, exhaustion, and a serious question about whether anyone was actually looking at my file.
5:08 PM — Automated call: APPROVED.
By 5:10 PM, I was not celebrating. I was shaking. I had screenshots of the approval text and denial texts. I had screenshots of each approval/denial in the patient portal. I called a live representative and asked if someone was dancing between the approval and denial buttons. I did not trust the portal — and for good reason.
That afternoon is a perfect illustration of what the PBM system looks like when its own processes break down under pressure. Nobody was coordinating. Automated systems were firing contradictory messages while human reviewers, regulators, and legal obligations were all pulling in different directions. They count on the chaos wearing you down until you stop asking. I kept asking.
What Finally Broke the Deadlock
The combination of three things ultimately resolved the denial: a properly documented appeal that directly addressed the denial criteria, a formal TDCI complaint that named specific Tennessee code violations, and persistent demands for a peer-to-peer review with a specialist — not a generalist.
Citing TN Code § 56-7-3504, held the PBM to step therapy regulations regardless of them claiming they did not define their policy in this case as step therapy. The PBM had referenced a drug "class" in their step therapy language. Under Tennessee law, that does not meet the legal standard for a valid step therapy requirement.
My doctor was my greatest ally throughout. Fighting a PBM alone is difficult. Fighting one alongside a physician who understands the legal landscape and is willing to push back is a completely different experience.
What I Want You to Take From This
I am sharing this not to describe how unusual my situation was, but to show how ordinary it is. This is how the system operates, not in spite of the rules, but often because of a willful disregard for them. Caremark paid $750,000 in penalties to the TDCI in early 2026 for repeated violations of Tennessee's insurance laws during 2024, including denying appeals without giving providers the legally required time to submit clinical information. (Source: Tennessee Department of Commerce and Insurance, February 2026.) This is a company that processes millions of prescriptions. The scale of what individual patients endure, quietly, every day, is staggering.
You are not powerless. You are, in fact, the one person with the most at stake, and that means you have every reason to become the most persistent, best-documented, legally-informed participant in this process.
A denial is not a diagnosis. It is not a medical opinion. It is a business decision — and business decisions can be challenged, documented, appealed, and reversed.
If you want the full tactical playbook for how to fight back at every level, read Beyond the Rejection: Power Moves for Overcoming Denials.




Comments